OVERVIEW
ABOUT PHARMACOGENOMICSPharmacogenomics (PGx) examines how genetic differences affect an individual’s response to medications. Variants in drug-related genes can influence how drugs are absorbed, metabolized, and cleared, which impacts both safety and efficacy. Research shows that up to 50% of adverse drug reactions (ADRs) may have a genetic cause1. PGx testing identifies genetic variants that influence drug response, enabling more precise, safer, and more effective treatment decisions, whether used proactively before treatment begins or in response to treatment failure or side effects.
PGx diagnostic testing includes a range of clinically focused tests designed to support treatment decisions in patients with a medical indication for therapy. These tests help identify genetic factors that may explain adverse drug reactions (ADRs), reduced efficacy, or dosing challenges by analyzing variants that affect drug metabolism, transport, and drug–target interactions.
Tests are structured by therapeutic area and focus on clinically relevant genes supported by CPIC3, DPWG4, and FDA5 guidelines. The results provide actionable insights for drug selection, dose optimization, management of complex or long-term therapies, and integration with therapeutic drug monitoring (TDM), where applicable.

Targeted tests designed to support treatment decisions in patients with a medical indication for therapy. Includes gene panels and single-gene analyses
tailored to relevant clinical specialties such as psychiatry, oncology, cardiology, and pain management.
WHO SHOULD TAKE THIS TEST?
Patients with unexplained side effects
or poor drug response
Individuals on high-risk medications
(e.g., chemotherapy, psychotropics)
Cases flagged by TDM or medication review
Patients with a clinical indication prior to
initiating high-risk treatment
1 ml EDTA blood or buccal swab
3-5 working days: PGx 5-Fluorouracil, PGx Irinotecan, PGx Mavacamten, PGx Siponimod
~ 1 week: PGx Azathioprine, PGx Paclitaxel
~ 2 weeks: PGx Eliglustat, PGx Tamoxifen
5-10 working days: PGx Abacavir, PGx Carbamazepine
10-15 working days: PGx Anesthesia and Pain, PGx Cardiovascular, PGx CYP450 Drug Response, PGx Metabolic Syndrome, PGx Neurology and Psychiatry, PGx Oncology
3-6 weeks: PGx Malignant Hyperthermia
Technologies include:
NGS-based amplicon sequencing (Illumina short-read + PacBio long-read)
PGx Anesthesia and Pain, PGx Cardiovascular, PGx CYP450 Drug Response, PGx Metabolic Syndrome, PGx Neurology and Psychiatry, PGx Oncology
NGS panel with CNV analysis (Illumina short-read)
PGx Malignant Hyperthermia
NGS-based amplicon sequencing (Illumina + PacBio) + SSO (sequence-specific oligonucleotides)
PGx Abacavir, PGx Carbamazepine
NGS-based amplicon sequencing (PacBio long-read)
PGx Eliglustat, PGx Tamoxifen
Quantitative real-time PCR (qPCR)
PGx 5-Fluorouracil, PGx Irinotecan, PGx Mavacamten, PGx Siponimod
NGS-based amplicon sequencing (Illumina short-read)
PGx Azathioprine, PGx Paclitaxel

POSSIBLE OUTCOMES OF THE TEST
PGx Diagnostic testing provides a comprehensive, clinically relevant report with clear and actionable insights based on the individual’s genetic profile.
The report includes a summary of the results, including variant details, therapeutic relevance and interpretation guidance.
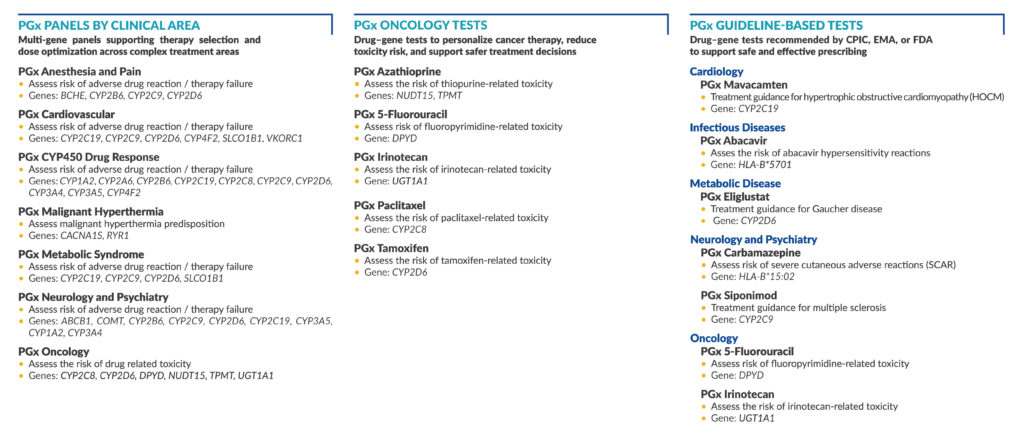
OUR TESTS
Genes: DYPD
Genes: HLA-B*5701
Genes: BCHE, CYP2B6, CYP2C9, CYP2D6
Genes: NUDT15, TPMT
Genes: HLA-B*15:02
Genes: CYP2C19, CYP2C9, CYP2D6, CYP4F2, SLCO1B1, VKORC1
Genes: CYP1A2, CYP2A6, CYP2B6, CYP2C19, CYP2C8, CYP2C9, CYP2D6, CYP3A4, CYP3A5, CYP4F2
Genes: CYP2D6
Genes: UGT1A1
Genes: CACNA1S, RYR1
Genes: CYP2C19
Genes: CYP2C19, CYP2C9, CYP2D6, SLCO1B1
Genes: BCB1, COMT, CYP2B6, CYP2C9, CYP2D6, CYP2C19, CYP3A5, CYP1A2, CYP3A4
Genes: CYP2C8, CYP2D6, DPYD, NUDT15, TPMT, UGT1A1
Genes: CYP2C8
Genes: CYP2C9
Genes: CYP2D6
Pharmacogenomic Preventive Panel is available to support safer, more personalized prescribing before treatment begins.
For customer service inquiries, please contact us at: info.genetics@medicover.com











