OVERVIEW
ABOUT PHARMACOGENOMICSPharmacogenomics (PGx) examines how genetic differences affect an individual’s response to medications. Variants in drug-related genes can influence how drugs are absorbed, metabolized, and cleared, which impacts both safety and efficacy. Research shows that up to 50% of adverse drug reactions (ADRs) may have a genetic cause1. By identifying these variants before treatment, PGx preventive screening supports more informed prescribing. It helps reduce side effects, improve treatment success, avoid ineffective or poorly tolerated medications, and lower healthcare costs by minimizing unnecessary medication adjustments.
The PGx Preventive Panel is a preemptive, patient-focused genetic test that supports safer and more personalized prescribing from the start. It analyzes key genetic variants that affect how drugs are absorbed, processed, and eliminated, helping to reduce side effects and improve treatment outcomes. The panel follows the latest CPIC3, DPWG4, and FDA5 recommendations. It is applicable across several areas of care, including pain management (e.g., NSAIDs, opioids), mental health (e.g., antidepressants, antipsychotics), cardiovascular care (e.g., statins, anticoagulants), and primary care/internal medicine (e.g., polypharmacy, geriatrics). Results are provided in a clear report with gene and drug interaction summaries and evidence-based dosing guidance to support informed clinical decisions.

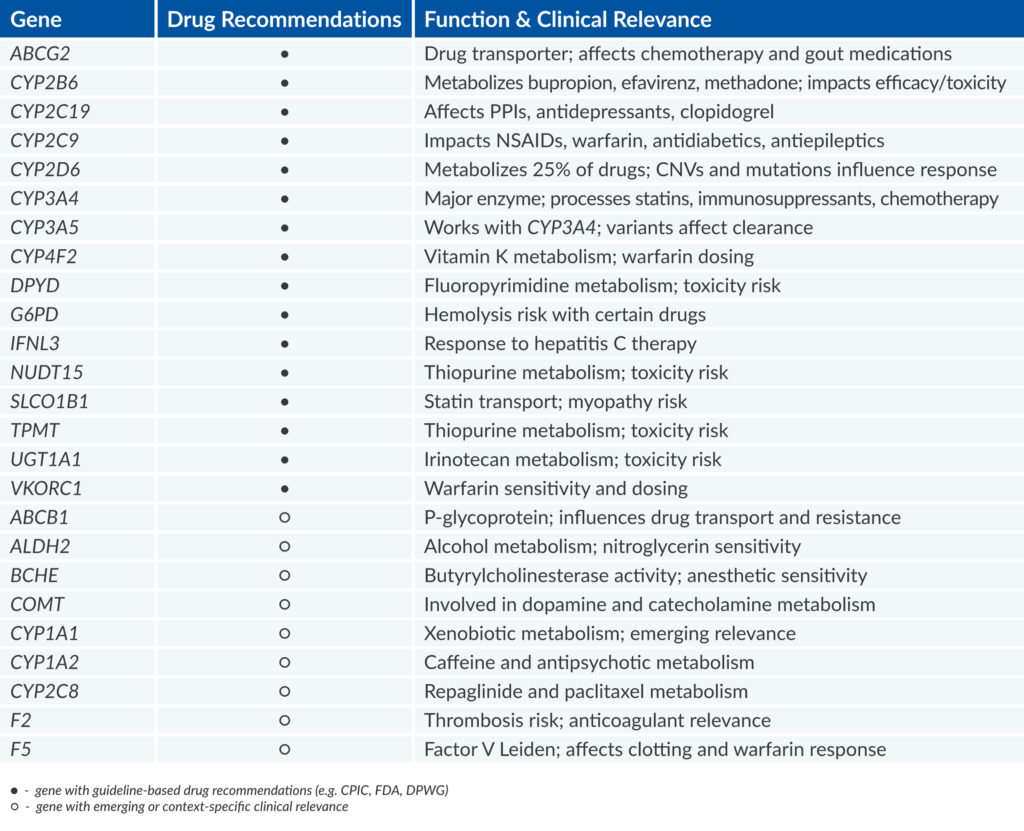
PGx Preventive panel analyzes 25 genes, including 16 core genes that are directly linked to drug response across more than 15 therapeutic areas.
These genes, such as CYP2D6, CYP2C19, CYP2C9, SLCO1B1, or TPMT, are involved in drug metabolism, transport, and response across multiple therapeutic areas,
and are supported by strong clinical evidence and high-level prescribing guidelines.
The panel is based on recommendations from leading pharmacogenomic societies3 and focuses on key genes involved in drug metabolism.
It includes a comprehensive analysis of the CYP2D6 gene, capturing full genotypes including critical copy number variations (CNVs) and hybrid alleles,
which are essential for accurate interpretation in clinical pharmacogenomics.
WHO SHOULD TAKE THIS TEST?
Patients interested in preventive and personalized healthcare
Patients seeking to optimize medication choices before potential health issues arise
Patients seeking to reduce the risk of ADRs or treatment failure
Patients managing multiple prescriptions (polypharmacy)

WHY RECOMMEND PGx PREVENTIVE PANEL?
Genetic variation accounts for up to 95% of how individuals respond to medications2. Over 100 drugs now include pharmacogenomic guidance in FDA labeling, showing the growing role of genetics in prescribing. Our PGx Preventive Panel provides clear, evidence-based results to guide drug and dose selection before issues arise.
It can help:
● Reduce the risk of ADRs
● Improve therapeutic efficacy
● Personalize treatment with greater precision
● Optimize long-term medication strategies
1 ml EDTA blood or buccal swab
10-15 working days
● Targeted short-read sequencing enables robust detection of actionable genotypes in clinically relevant PGx genes
● CYP2D6 analysis via long-read sequencing resolves complex structural variants in the CYP2D6-CYP2D7 locus, identifying gene duplications, deletions, and hybrid genes that may be missed by standard methods
● Guideline-aligned interpretation based on international pharmacogenomic standards, including CPIC, DPWG, and FDA recommendations

POSSIBLE OUTCOMES OF THE TEST
The PGx Preventive panel provides a comprehensive, clinically relevant report with clear and actionable insights based on the individual’s genetic profile.
The report includes a summary of key gene-drug interactions relevant to the individual. A total of 25 genes are analyzed, including 16 key pharmacogenes involved in drug metabolism. Star alleles are detected and reported to support an accurate interpretation of the patient’s profile.
For the 16 core pharmacogenes, the report offers precise dose recommendations based on the patient’s genotype. Each gene is annotated with its clinical significance, mode of action, and potential implications. The report also includes interpretation guidance, relevant disclaimers, and a comprehensive list of scientific references to support the recommendations.
Pharmacogenomic Diagnostic Testing is available for patients with a confirmed diagnosis, supporting more targeted and effective treatment decisions.
For customer service inquiries, please contact us at: info.genetics@medicover.com











