Thyroid carcinomas are the most common endocrine tumors and are responsible for 1% to 5% of tumors in women and <2% in men. It is a heterogeneous disease that originates from two different epithelial cell types. Most thyroid carcinomas develop from follicular cells (follicular cell derived thyroid carcinoma). These include papillary (PTC; 65-80%), follicular (FTC; 10-30%), Hürthle cell and poorly differentiated (OV-/PDTC; <2%) and anaplastic thyroid carcinomas (ATC; <2%). PTC and FTC together are called differentiated thyroid carcinomas (DTC) and account for 90% to 95% of all thyroid carcinomas. In general, these are slow-growing tumors with an excellent prognosis: 20-year survival rates are >90% with conventional therapy consisting of resection with or without thyroidectomy with radioactive iodine therapy and suppressive thyroid hormone therapy.
GENOMIC ALTERATIONS AND POSSIBLE THERAPIES
In the majority of cases, DTC is caused by molecular changes in so-called driver genes such as BRAF and RAS or by the creation of fusion genes. These changes lead to the pathological activation of the MAPK signaling pathway, which plays a key role in the regulation of many cellular processes. By accumulating further molecular genetic alterations, e.g., in TERT, tumor suppressor genes, or the phosphoinositide-3-kinase signaling pathway, DTC can change into dedifferentiated PDTC or ATC. However, a proportion of patients with DTC show more aggressive disease progression with a recurrent or metastatic course and an insensitivity to radioiodine therapy.
Medullary thyroid carcinomas (MTC) develop from parafollicular calcitonin-producing cells and can be part of multiple endocrine neoplasia type 2 (MEN2), a hereditary disposition for the development of MTC.
The papillary thyroid carcinoma (PTC) is by far the most common entity within the group of epithelial thyroid carcinoma with about 65% to 80%. It is characterized by chromosomal rearrangements involving the RET gene (RET/PTC rearrangements or RET fusion genes) as well as pathogenic variants within the RAS or BRAF proto-oncogenes, all of which ultimately lead to pathological activation of the MAPK signaling pathway. Variants in the BRAF (Val600Glu, Lys601Glu), NRAS (codon 12, 13, and 61), HRAS, or RET genes are found in almost 70% of all cases of PTC.
Tumors with variants in BRAF are usually undifferentiated and associated with more aggressive tumor characteristics such as a poorer response to radioactive iodine therapy. BRAF Val600Glu is found in 45% to 59% of PTC, primarily in the classical papillary and large cell variant and in up to 80% of patients with recurrent or metastatic PTC.
PTC with variants in NRAS, HRAS, and KRAS are more frequently differentiated and are found in 10-20% of the follicular variant of PTC. Pathogenic variants in BRAF and RAS are almost never found together in PTC tumors. Pathogenic variants in the TERT promoter (Cys288Thr; Cys250Thr) are also found in up to 10% of all PTC. They are associated with a poorer clinical outcome, especially in combination with BRAF variants.
Follicular thyroid carcinoma (FTC) is the second most common entity (10-30%). FTC has a different variant profile with a relatively higher proportion of pathogenic RAS variants (about 30-40% of all FTCs), whereas BRAF Val600Glu and other BRAF variants are rarely found (0-4%). Interestingly, changes are relatively frequent in another candidate driver gene, EIF1AX (5-7%). In addition, TERT variants are also found in FTC and seem to be associated with a significantly worse prognosis.
Medullary thyroid carcinomas (MTC) account for about 3% to 5% of thyroid carcinomas. About 25% of MTC are hereditary and show germline variants in the RET gene. In addition, somatic variants can also be detected in RET in up to 66% of sporadic MTC. In 75% to 95% of the cases, a RET Met918Thr variant is found, which is associated with a more aggressive course, as is the rarer Ala883Phe variant (high-risk variants). The simultaneous occurrence of several somatic variants in the RET gene is also associated with a worse prognosis. In up to 81% of sporadic, RET wild-type MTC, variants are found in HRAS, especially Gln61Arg, and less frequently in KRAS. The role of Ras variants is less well characterized, but there are data that suggest tumors with somatic RAS variants are less aggressive than those with high-risk RET variants but more aggressive than those with non-high-risk RET variants. The mTOR signal pathway is also activated, both in hereditary and sporadic MTC. There is evidence that high mTOR activity is associated with more invasive sporadic MTC and a higher rate of lymph node metastasis.
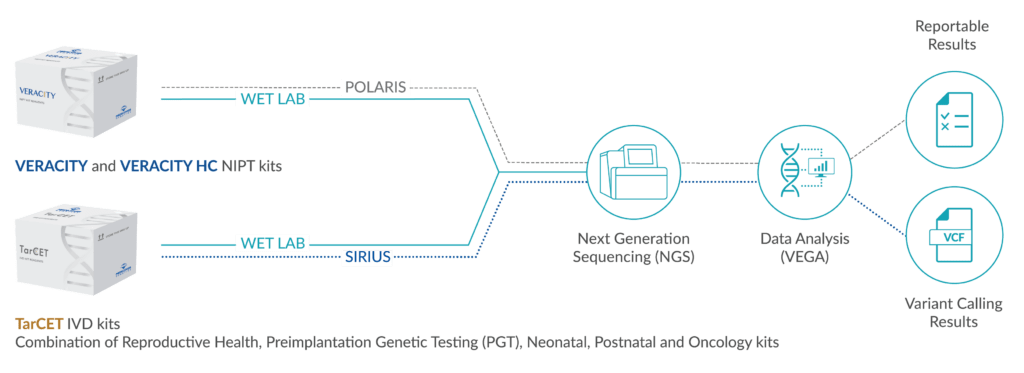
GENE PANEL
7 genes: BRAF, EIF1AX, HRAS, KRAS, NRAS, RET, TERT
FUSION GENE PANEL
AKAP9-BRAF, CCDC6-RET, CRTC1-MAML2, EML4-ALK, ERC1-RET, ETV6-NTRK3, GOLGA5-RET, HOOK3-RET, NCOA4-RET, NTRK1-TFG, NTRK1-TPM3, PAX8-PPARG, PCM1-RET, PRKAR1A-RET, RET-GOLGA5, STRN-ALK, TFG-NTRK1, TPM3-NTRK1, TRIM24-RET, TRIM27-RET, TRIM33-RET