OVERVIEW
DETECTClose to 20 million new cancer cases and 10 million deaths have occurred annually in recent years. Solid tumors represent approximately 90% of adult cancers, and millions of histopathology slides are analyzed each year, providing crucial information for cancer diagnosis and staging. In up to 40% of patients, complex genomic alterations are identified, which can serve as biomarkers to predict response to specific therapies and/or prognosis.
Histopathological examination and genetic testing can determine the tumor profile and guide management and treatment plans.
Characterizing the cellular and molecular changes in a solid tumor is critical for determining the treatment strategy, as customized treatment depends on the type, severity (stage), and specific genetic alterations in the tumor tissue. Genotype-directed therapy or genotype-matched clinical trials can significantly improve patient care and survival.

IMPORTANCE OF GETTING TESTED
Histopathology and genetic tumor profiling allow for individualized and more efficient and personalized treatment. Treating a tumor based on its unique cellular and molecular features may result in a better outcome, including improved clinical management and increased survival.

Patients who are diagnosed with
a solid tumor and require
a pathological and/or genetic test analysis
OUR SOLUTION
We provide both histopathology and molecular genetic analyses, with the flexibility to select either service individually or to combine them for a more comprehensive approach to diagnosis and treatment planning.
HISTOPATHOLOGY
Microscopic examination of the tumor sample provides specific information about the type and the stage of the tumor, its location and size and if it has spread to other healthy parts of the body.
GENETIC TESTS
Based on the histopathological findings, genetic analysis may be recommended. Your physician can choose from individual targeted-therapy tests, gene panels or a single comprehensive gene panel.
Consider Hereditary Cancer Panels Predict&Prevent if your patient’s tumor has a genetic cause


TEST OPTIONS
GENETIC TESTING only
● Comprehensive Panel
● Targeted-Therapy Tests
By combining histopathology and genetic testing, you can receive a complete diagnosis of the tumor which will guide your physician to decide on the most appropriate treatment for you, as well as recommend clinical trials that can help improve the clinical outcome.
Identifies specific genomic changes that are relevant to therapy with approved therapeutic products (targeted therapies)

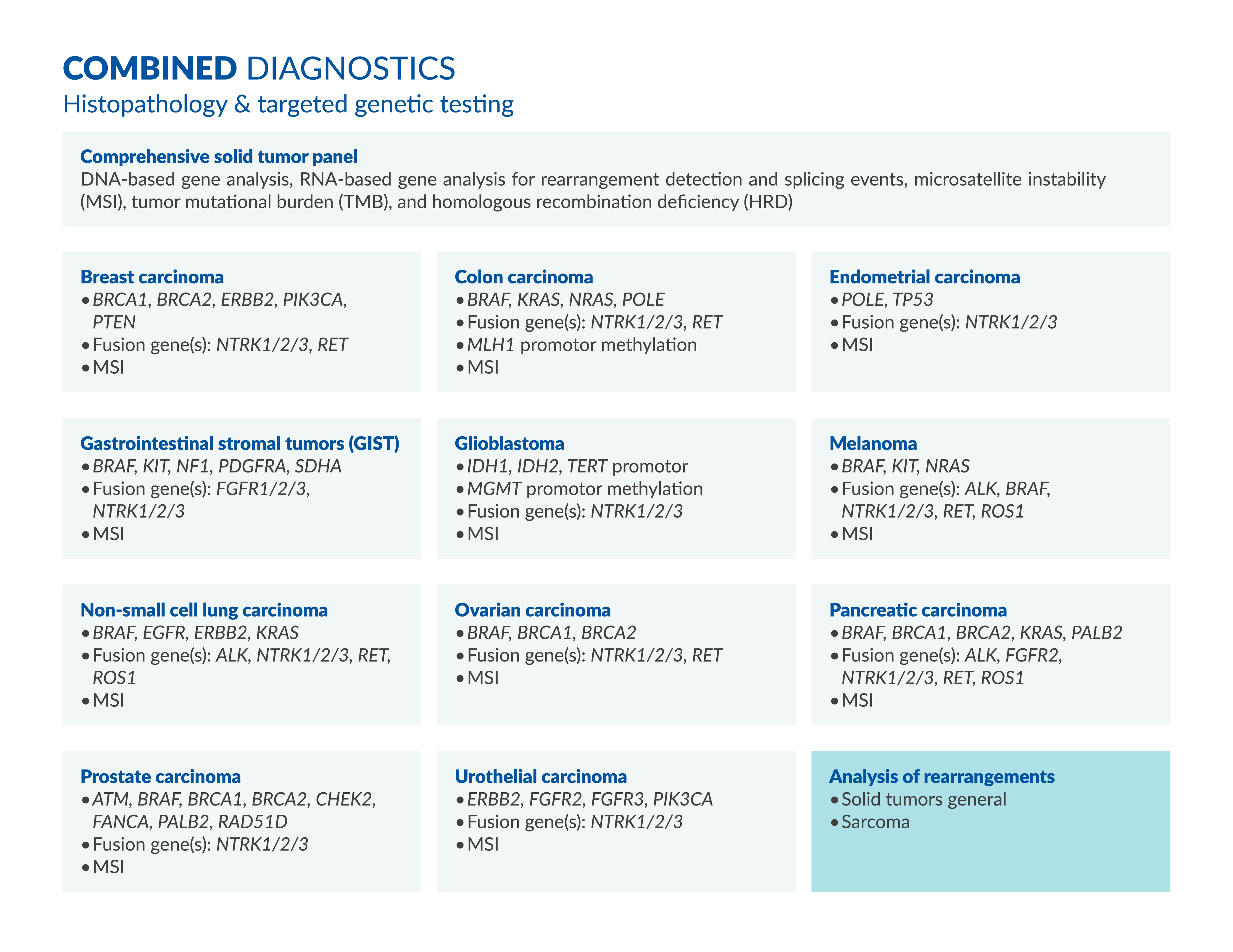
DNA-based gene analysis, RNA-based gene analysis for rearrangement detection and splicing events, microsatellite instability (MSI), tumor mutational burden (TMB), and homologous recombination deficiency (HRD)
• Solid tumors general
• Sarcoma

POSSIBLE OUTCOMES OF THE TEST
A diagnostic report outlining the results of the histopathology and/or the sequencing results from genetic testing is provided.
Pathology report includes macroscopic and microscopic descriptions of the provided sample, results of all tests conducted, and conclusions based on the final histopathological diagnosis. Conclusions include classification of lesions, WHO disease code, and recommendations, with the suggestion to perform sequencing analyses included if necessary.
Sequencing report includes changes in DNA sequence (variants) including single nucleotide variants, copy number variants (only for comprehensive panel) and fusion genes, as well as tumor mutational burden and microsatellite instability, as well as the effect on carcinogenesis. Possible therapy options and clinical trials relevant to the patient’s location will be listed.
Targeted-therapy analysis, solid tumor panels and the comprehensive tumor panel are covered as part of the sequencing report where applicable.
Targeted-therapy analysis identifies specific genomic changes that are relevant to therapy with approved therapeutic products (targeted therapies).
Solid tumor panels and comprehensive tumor panel may identify additional genomic findings that are not prescriptive or conclusive for use of any targeted therapies. Use of these panels does not guarantee a patient will be matched to a treatment. A negative result does not rule out the presence of an alteration.
MEDICAL GENETIC COUNSELLING
We provide expert medical genetic counselling as part of a genetic testing journey. Genetic counselling is a process of communication that supports patients and their relatives before and after genetic testing. It is educational, impartial and nondirective. Prior to any genetic test, genetic counsellors will obtain a detailed family history, explain the method of testing that will be used, its risks and benefits, the limitations of the diagnosis and the implications of making a genetic diagnosis (Elliott and Friedman, 2018, Nat Rev Genet 19:735).
Upon receiving the genetic test results, genetic counselling can help the specialist physician and the patient to interpret them. They can be advised of the consequences of the results including the probability of developing the genetic disorder or passing it on to children, as well as ways to prevent, avoid or reduce these risks (Yang and Kim, 2018, Ann Lab Med 38:291). Our goal of counselling is to provide the patient with greater knowledge and thus, a better understanding of the results and the ability to make a more informed decision.

Tissue fragments from a biopsy:
– Stored in 10% formalin (stable for 24-72 hrs)
– Embedded in paraffin blocks (stored in dry and dark conditions)
Histopathology analysis: 7-10 working days
Genetic analysis: 7-20 working days
Information updated soon
OUR TESTS
Genes: BRAF, BRCA1, BRCA2, Fusion Genes: NTRK1, NTRK2, NTRK3, RET
Genes: BRAF, KRAS, NRAS, POLE, Fusion Genes: NTRK1, NTRK2, NTRK3, RET
Genes: 638 DNA-based genes, 22 RNA-based genes DNA: ABL1, ABL2, ACVR1, ACVR1B, ADARB2, AGO1, AGO2, AJUBA, AKT1, AKT2, AKT3, ALB, ALK, ALOX12B, AMER1, ANKRD11, ANKRD26, APC, APLNR, AR, ARAF, ARFRP1, ARHGAP35, ARID1A, ARID1B, ARID2, ARID5B, ASXL1, ASXL2, ATM, ATR, ATRX, ATXN7, AURKA, AURKB, AXIN1, AXIN2, AXL, B2M, BABAM1, BAP1, BARD1, BBC3, BCL10, BCL2, BCL2L1, BCL2L11, BCL2L2, BCL6, BCOR, BCORL1, BCR, BIRC3, BLM, BMPR1A, BRAF, BRCA1, BRCA2, BRD4, BRIP1, BTG1, BTG2, BTK, CALR, CARD11, CARM1, CASP8, CBFB, CBL, CCNB3, CCND1, CCND2, CCND3, CCNE1, CD276, CD70, CD74, CD79A, CD79B, CDC42, CDC73, CDH1, CDH4, CDK12, CDK4, CDK6, CDK7, CDK8, CDKN1A, CDKN1B, CDKN2A, CDKN2B, CDKN2C, CEBPA, CENPA, CHD2, CHD4, CHEK1, CHEK2, CIC, CMTR2, CNTN4, CREBBP, CRKL, CRLF2, CSDE1, CSF1R, CSF3R, CSNK1A1, CTCF, CTLA4, CTNNA1, CTNNB1, CTR9, CUL3, CUL4A, CUX1, CXCR4, CYLD, CYP17A1, CYP19A1, CYP2C19, CYP2D6, CYSLTR2, DAXX, DCUN1D1, DDR1, DDR2, DDX41, DHX15, DICER1, DIS3, DNAJB1, DNMT1, DNMT3A, DNMT3B, DOT1L, DPYD, DROSHA, DUSP4, E2F3, EED, EGFL7, EGFR, EIF1AX, EIF4A2, EIF4E, ELF3, EML4, EMSY, EP300, EPAS1, EPCAM, EPHA3, EPHA5, EPHA7, EPHB1, EPHB4, ERBB2, ERBB3, ERBB4, ERCC1, ERCC2, ERCC3, ERCC4, ERCC5, ERF, ERG, ERRFI1, ESR1, ETAA1, ETS1, ETV1, ETV4, ETV5, ETV6, EWSR1, EZH1, EZH2, EZR, FAM175A, FAM46C, FAM58A, FANCA, FANCC, FANCD2, FANCE, FANCF, FANCG, FANCI, FANCL, FAS, FAT1, FBXW7, FGF1, FGF10, FGF12, FGF14, FGF19, FGF2, FGF23, FGF3, FGF4, FGF5, FGF6, FGF7, FGF8, FGF9, FGFR1, FGFR2, FGFR3, FGFR4, FH, FLCN, FLI1, FLT1, FLT3, FLT4, FOXA1, FOXF1, FOXL2, FOXO1, FOXP1, FRS2, FUBP1, FYN, GAB1, GAB2, GABRA6, GATA1, GATA2, GATA3, GATA4, GATA6, GEN1, GID4, GLI1, GNA11, GNA13, GNAQ, GNAS, GNB1, GPR124, GPS2, GREM1, GRIN2A, GRM3, GSK3B, H3F3A, H3F3B, H3F3C, HDAC1, HGF, HIST1H1C, HIST1H2BD, HIST1H3A, HIST1H3B, HIST1H3C, HIST1H3D, HIST1H3E, HIST1H3F, HIST1H3G, HIST1H3H, HIST1H3I, HIST1H3J, HIST2H3A, HIST2H3C, HIST2H3D, HIST3H3, HLA-A, HLA-B, HLA-C, HNF1A, HNRNPK, HOXB13, HRAS, HSD3B1, HSP90AA1, ICOSLG, ID3, IDH1, IDH2, IFNGR1, IGF1, IGF1R, IGF2, IKBKE, IKZF1, IL10, IL7R, INHA, INHBA, INPP4A, INPP4B, INPPL1, INSR, IRF2, IRF4, IRS1, IRS2, JAK1, JAK2, JAK3, JUN, KAT6A, KBTBD4, KDM5A, KDM5C, KDM6A, KDR, KEAP1, KEL, KIF5B, KIT, KLF4, KLF5, KLHL6, KMT2A, KMT2B, KMT2C, KMT2D, KMT5A, KNSTRN, KRAS, LAMP1, LATS1, LATS2, LMO1, LRP1B, LTK, LYN, LZTR1, MAD2L2, MAGI2, MALT1, MAP2K1, MAP2K2, MAP2K4, MAP3K1, MAP3K13, MAP3K14, MAP3K4, MAPK1, MAPK3, MAPKAP1, MAX, MCL1, MDC1, MDM2, MDM4, MED12, MEF2B, MEN1, MET, MGA, MITF, MLH1, MLLT1, MLLT3, MPL, MRE11A, MSH2, MSH3, MSH6, MSI1, MSI2, MST1, MST1R, MTAP, MTOR, MUTYH, MYB, MYC, MYCL, MYCN, MYD88, MYOD1, NAB2, NADK, NBN, NCOA3, NCOR1, NEGR1, NF1, NF2, NFE2L2, NFKBIA, NKX2-1, NKX3-1, NOTCH1, NOTCH2, NOTCH3, NOTCH4, NPM1, NRAS, NRG1, NSD1, NT5C2, NTHL1, NTRK1, NTRK2, NTRK3, NUF2, NUP93, NUTM1, OPCML, P2RY8, PAK1, PAK3, PAK7, PALB2, PARK2, PARP1, PARP2, PARP3, PAX3, PAX5, PAX7, PAX8, PBRM1, PD-1, PD-L1, PD-L2, PDGFRA, PDGFRB, PDK1, PDPK1, PGBD5, PGR, PHF6, PHOX2B, PIGA, PIK3C2B, PIK3C2G, PIK3C3, PIK3CA, PIK3CB, PIK3CD, PIK3CG, PIK3R1, PIK3R2, PIK3R3, PIM1, PLCG2, PLK2, PMAIP1, PMS1, PMS2, PNRC1, POLD1, POLE, POT1, PPARG, PPM1D, PPP2R1A, PPP2R2A, PPP4R2, PPP6C, PRDM1, PRDM14, PREX2, PRKAR1A, PRKCI, PRKD1, PRKDC, PRSS8, PTCH1, PTEN, PTP4A1, PTPN11, PTPRD, PTPRN2, PTPRS, PTPRT, QKI, RAB35, RAC1, RAC2, RAD21, RAD50, RAD51, RAD51B, RAD51C, RAD51D, RAD52, RAD54L, RAF1, RANBP2, RARA, RASA1, RB1, RBM10, RECQL, RECQL4, REL, REST, RET, RFWD2, RHEB, RHOA, RICTOR, RIT1, RNF43, ROS1, RPS6KA4, RPS6KB1, RPS6KB2, RPTOR, RRAGC, RRAS, RRAS2, RSPO2, RTEL1, RUNX1, RUNX1T1, RXRA, RYBP, SCG5, SDC4, SDHA, SDHAF2, SDHB, SDHC, SDHD, SERPINB3, SERPINB4, SESN1, SESN2, SESN3, SETBP1, SETD2, SETDB1, SF3B1, SGK1, SH2B3, SH2D1A, SHOC2, SHQ1, SLC34A2, SLFN11, SLIT2, SLX4, SMAD2, SMAD3, SMAD4, SMARCA2, SMARCA4, SMARCB1, SMARCD1, SMARCE1, SMC1A, SMC3, SMO, SMYD3, SNCAIP, SNTG2, SOCS1, SOS1, SOX10, SOX17, SOX2, SOX9, SPEN, SPOP, SPRED1, SPRTN, SPTA1, SRC, SRSF2, STAG1, STAG2, STAT3, STAT4, STAT5A, STAT5B, STK11, STK19, STK40, SUFU, SUZ12, SYK, TAF1, TAP1, TAP2, TBX3, TCEB1, TCF3, TCF7L2, TEK, TERT, TET1, TET2, TFE3, TFRC, TGFBR1, TGFBR2, TIPARP, TMEM127, TMPRSS2, TNFAIP3, TNFRSF14, TOP1, TOP2A, TP53, TP53BP1, TP63, TPMT, TRAF2, TRAF7, TRIP13, TSC1, TSC2, TSHR, TYRO3, U2AF1, UGT1A1, UPF1, USP8, VEGFA, VHL, VTCN1, WHSC1, WHSC1L1, WISP3, WT1, WWTR1, XIAP, XPO1, XRCC2, YAP1, YES1, ZBTB2, ZBTB7A, ZFHX3, ZNF217, ZNF703, ZNRF3, ZRSR2 RNA: ALK, ROS1, RET, FGFR1, FGFR2, FGFR3, NTRK1, NTRK2, NTRK3, BRAF, NRG1, EWSR1, TMPRSS2, BRCA1, BRCA2, PTEN, AR, EGFR, ERBB2, MET, PALB2, RB1
Genes: POLE, TP53, Fusion Genes: NTRK1, NTRK2, NTRK3, MSI
Genes: BRAF, KIT, NF1, PDGFRA, SDHA, Fusion Genes: NTRK1, NTRK2, NTRK3
Genes: IDH1, IDH2, TERT promotor, MGMT promotor methylation, Fusion Genes: NTRK1, NTRK2, NTRK3
Genes: RAF, KIT, NRAS, Fusion Genes: ALK, BRAF, NTRK1/2/3, RET, ROS1
Genes: BRAF, EGFR, ERBB2, KRAS, Fusion Genes: ALK, NTRK1/2/3, RET, ROS1
Genes: BRCA1, BRCA2, ERBB2, PIK3CA, PTEN, Fusion Genes: NTRK1, NTRK2, NTRK3, RET
Genes: BRAF, BRCA1, BRCA2, KRAS, PALB2, Fusion Genes: ALK, FGFR2, NTRK1, NTRK2, NTRK3, RET, ROS1
Genes: ATM, BRAF, BRCA1, BRCA2, CHEK2, FANCA, PALB2, RAD51D, Fusion Genes: NTRK1, NTRK2, NTRK3
Genes: Fusion Genes: NTRK3::ETV6, EWSR1::NR4A3, EWSR1::PBX1, EWSR1::ZNF384, EWSR1::ATF1, EWSR1::PATZ1, EWSR1::DDIT3, EWSR1::SP3, EWSR1::FEV, EWSR1::CREB1, EWSR1::FLI1, EWSR1::ETV4, EWSR1::ETV1, EWSR1::ERG, YY1::EWSR1, EWSR1::ZNF444, EWSR1::SMARCA5, NFATC2::EWSR1, SS18::SSX1, SS18::SSX4, FUS::CREB3L2, FUS::CREB3L1, FUS::DDIT3, FUS::ERG, FUS::ATF1, FUS::FEV, PAX3::FOXO1, PAX7::FOXO1
Genes: Fusion Genes: A2M::ALK, ACTG2::ALK, ALK::PTPN3, ATIC::ALK, C2orf44::ALK, CARS::ALK, CLIP4::ALK, CLTC::ALK, DCTN1::ALK, EML4::ALK, ETV6::ALK, GTF2IRD1::ALK, HIP1::ALK, KIF5B::ALK, KLC1::ALK, LMNA::ALK, MEMO1::ALK, MPRIP::ALK, MSN::ALK, NCOA1::ALK, PPFIBP1::ALK, PPP4R3B::ALK, PRKAR1A::ALK, RANBP2::ALK, SEC31A::ALK, STRN::ALK, SQSTM1::ALK; TFG::ALK, TPM1::ALK, TPM3::ALK, TPM4::ALK, TPR::ALK, TRAF1::ALK, VCL::ALK, ACBD5::RET, AFAP1::RET, AKAP13::RET, CCDC6::RET, CUX1::RET, ERC1::RET, FKBP15::RET, GOLGA5::RET, HOOK3::RET, KIAA1468::RET, KIF5B::RET, KTN1::RET, MYO5A::RET, NCOA4::RET, PCM1::RET, PRKAR1A::RET, RUFY2::RET, SPECC1L::RET, SQSTM1::RET, TBL1XR1::RET, TFG::RET, TRIM24::RET, TRIM27::RET, TRIM33::RET, CD74::ROS1, CEP85L::ROS1, CCDC6::ROS1, CLIP1::ROS1, CLTC::ROS1, ERC1::ROS1, EZR::ROS1, GOPC::ROS1, HLA-A::ROS1, KDELR2::ROS1, LRIG3::ROS1, MSN::ROS1, MYO5A::ROS1, PPFIBP1::ROS1, PWWP2A::ROS1, SDC4::ROS1, SEC34A2::ROS1, SHTN1::ROS1, TFG::ROS1, TPM3::ROS1, ZCCHC8::ROS1, BCAN::NTRK1, CD74::NTRK1, CEL::NTRK1, IRF2BP2::NTRK1, LMNA::NTRK1, MPRIP::NTRK1, NFASC::NTRK1, NTRK1::DYNC2H1, RNF213::NTRK1, SQSTM1::NTRK1, SSBP2::NTRK1, TFG::NTRK1, TPM3::NTRK1, TPR::NTRK1, AFAP1::NTRK2, AGBL4::NTRK2, NACC2::NTRK2, QKI::NTRK2, SQSTM1::NTRK2, TRIM24::NTRK2, VCL::NTRK2, BTBD1::NTRK3, COX5A::NTRK3, ETV6::NTRK3L
Genes: ERBB2, FGFR2, FGFR3, PIK3CA, Fusion Genes: NTRK1, NTRK2, NTRK3











